Portable Cancer Diagnostic Tool
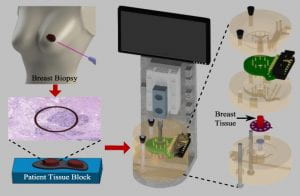
The objective of this study is to design and develop a portable tool consisting of a disposable biochip for measuring electro-thermo-mechanical (ETM) properties of breast tissue. A biochip integrated with a microheater, force sensors, and electrical sensors is fabricated using microtechnology. A portable tool capable of holding tissue and biochip is fabricated using 3-D printing. Invasive ductal carcinoma and normal tissue blocks are selected from cancer tissue bank in Biospecimen Repository Service at Rutgers Cancer Institute of New Jersey. The ETM properties of the normal and cancerous breast tissues (3 mm thickness and 2 mm diameter) are measured by indenting the tissue placed on the biochip integrated inside the 3-D printed tool. Integrating microengineered biochip and 3-D printing, we have developed a portable cancer diagnosis device. Using this device, we have shown a statistically significant difference between cancerous and normal breast tissues in mechanical stiffness, electrical resistivity, and thermal conductivity. Conclusion: The developed cancer diagnosis device is capable of simultaneous ETM measurements of breast tissue specimens and can be a potential candidate for delineating normal and cancerous breast tissue cores. The portable cancer diagnosis tool could potentially provide a deterministic and quantitative information about the breast tissue characteristics, as well as the onset and disease progression of the tissues. The tool can be potentially used for other tissue-related cancers.
Machine Learning Approach for Cancer Localization
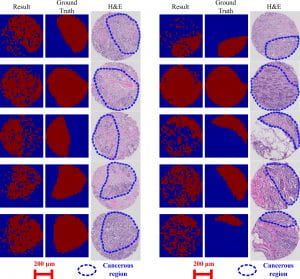
Machine learning has been widely used for solving various classification problems in biomedical research field due to its strength in handling massive data set systematically. In this paper, a feasible way of breast cancer localization via machine learning is presented with a preliminary result for 10 cancerous breast tissue samples (600 µm diameter and 8 µm thickness). Using a custom-built microscope-compatible microindentation system, 500 indentation points per sample were indented to a depth of 2 µm at 20 µm interval. Each indentation point is labeled as either ’Normal’ or ’Cancerous’ according to the corresponding pathological image that was annotated appropriately by a certified pathologist. We applied the support vector machine (SVM) algorithm, which is one of the supervised machine learning technique to validate the annotations by a pathologist. The tissue elasticity value which is the actual data set for machine learning is locally determined by a non-linear contact model for a spherical tip, using the contact force and the indentation depth information collected during the indentation experiment. With soft-margin SVM which is for not linearly separable data, each tissue sample is tested while the other 9 tissue samples are used for training like 10-fold cross-validation. Classification accuracy for the entire breast tissue samples was obtained as 76.20% ± 9.28%, which shows that this is a promising approach when making allowance for classification with a single parameter.
Mechano-Visual Phenotyping of Breast Cancer

The goal of this project is to develop experimental and computational tools to characterize the onset and progression of cancer in human breast tissue. We use contact-mode Atomic Force Microscopy (AFM) to characterize the spatial distribution of the elastic properties of benign and cancerous breast tissue specimens. To increase characterization throughput across specimens larger than the travel range of commercial AFM scanners, we have developed a customized image-guided positioning system capable of aligning the tissue specimens with the AFM across multiple magnifications. In addition, we have also developed mathematical models to quantify characterization errors arising due to the physical limits of the AFM instrumentation and calibration uncertainties.
MEMS-based Breast Cancer Diagnosis
Mechanical Characterization of Benign and Cancerous Breast Tissue using MEMS Sensor

The goal of this project is to develop mechanical characterization technique for benign and cancerous breast tissue in a high throughput manner. Contact-mode Atomic Force Microscopy (AFM), which is popularly used in quantifying material properties of biomaterials, has limitations like complex read-out electronics, bulky optics, and inability to use in opaque liquids. To overcome these problems, we have designed and developed Micro-Electro-Mechanical-System (MEMS) based Piezoresistive microcantilever force sensors with a cylindrical tip made from SU-8 polymer. These force sensors are used for detecting cancer progression in breast tissue. An array of piezoresistive microcantilever beam is envisaged to improve throughput and will be a cost-effective approach for quantifying the mechanical properties of the benign and cancerous breast tissues.
Electrical Characterization of Benign and Cancerous Breast Tissue using MEMS based Microchips
 The goal of this project is to build an automated system using MEMS-baseddevices capable of measuring impedance of benign or cancerous breast tissue array simultaneously, thus increasingthe sensing throughput. We have designed and fabricated microchips having interdigital electrodesinside a SU-8 well to measure the impedance of benign and cancerous breast tissues. The benign and cancerous breast tissue core is placed in the center of the interdigital electrodes and the bio-impedance is measured. From theexperiments conducted, we found that cancerous breasttissue specimens displayed significantly different bio-impedance characteristics compared tobenign breast tissue specimens. We plan to integrate bio-impedance measurement with mechanicalcharacterization for automated sampling of breast tissue core specimens.
The goal of this project is to build an automated system using MEMS-baseddevices capable of measuring impedance of benign or cancerous breast tissue array simultaneously, thus increasingthe sensing throughput. We have designed and fabricated microchips having interdigital electrodesinside a SU-8 well to measure the impedance of benign and cancerous breast tissues. The benign and cancerous breast tissue core is placed in the center of the interdigital electrodes and the bio-impedance is measured. From theexperiments conducted, we found that cancerous breasttissue specimens displayed significantly different bio-impedance characteristics compared tobenign breast tissue specimens. We plan to integrate bio-impedance measurement with mechanicalcharacterization for automated sampling of breast tissue core specimens.
